Plant cells
Biological description
- Cell walls in land plants are primarily composed of cellulose microfibrils embedded in a pectin and hemicellulose matrix (Noguchi 2014, 137). The secondary walls of plant cells are structurally rigid and contain three dense layers of cellulose and hemicellulose arranged in microfibrils of varying orientations that promote strength (Evert 2006a, 74; Raven et al. 1999, 65). The layered substructure of the cell wall and microfibrils are not visible when viewing in situ residues with reflected light microscopy, but have been characterised by atomic force microscopy (Kirby et al. 1996; Morris et al. 1997; Radotić et al. 2008). Cell walls and stomata are very resilient structures, which is supported by evidence that suggests they can survive hostile burial environments. In fact, cell walls sometimes still survive after acetolysis, a standard palynological processing technique (Moore and Webb 1978, 24) that is meant to digest and remove cellulose and other organics from pollen preparations.
Identification
- Although there is significant variability both in morphology and size, plant tissue cells appear distinctive under the microscope: their walls always have a clear bounded structure. The reed, softwood, and hardwood tissues all exhibited recognisable cell walls.
- More specific identification beyond 'plant tissue cells' is sometimes possible with VLM; however, features in the cell wall such as pits, elaborations of the secondary wall, or specialised structures must be visible.
Reference residue
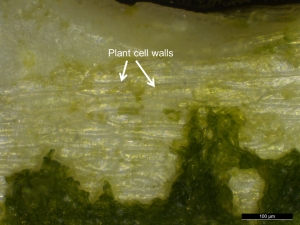
- The residues from reeds contained cells with an elongate sub-rectangular structure and well-defined cell walls (Figure 29).
- Green chlorophyll pigments within the reed residue were also still intact at the time of imaging, although these would not be expected to survive on archaeological specimens.
- Examination of the reed residues with SEM illustrated cell wall structure and also showed the reeds contain long crystals (Figure 30).

Starch
Biological description
- Green plants store the glucose products of photosynthesis as amylose and amylopectin, which together comprise starch granules (Raven et al. 1999; Simpson 2010).
- There are three types of starch that occur in plants: small temporary or transient starches found in the chloroplasts, large statolith granules found in the root cap cells used for perception of gravity, and storage starch, found in a variety of plant tissues – the latter type being of greatest use to archaeologists.
- Starch granule structure is controlled by genetics, developmental stage of the plant, and environmental factors.
Identification
- Starch grains are clear, usually colourless and have a mean refractive index of 1.5, which is somewhat affected by moisture content (Blanshard 1987, 19). Important characteristics of starch granule morphology are: birefringence, the presentation and rotation of an extinction cross, three-dimensional shape, hilum (open or closed, centric or eccentric location, colour), lamellae (visible or invisible), fissures (present or absent, size), simple (occuring singly) versus compound granules (aggregates), and facets (present or absent).
Identification notes
- Starch granules in situ on stone surfaces cannot be diagnostically identified with reflected VLM, but their presence may be suggested by size, shape, and display of extinction crosses.
- Extinction crosses of undamaged starch granules are sometimes visible during in situ viewing with cross polarised light. However, the presence of an extinction cross within a round or oval shape is not diagnostic of starch alone, as many microscopic items present extinction crosses, including mineral and coprolitic spherulites (Canti 1998; 1999), conidia fungal spores (Haslam 2006b), bordered pits on plant tracheids and vessel elements (Wayne 2009), phytoplankton and zooplankton with calcareous shells including coccoliths (Loy 2006), bivalve veligers (larvae), ostracods and echinoderm larvae (Johnson 1995). Other features must be present in conjunction with the presentation of an extinction cross whose arms rotate when the polariser is rotated.
- Starch is more readily identifiable with a transmitted light microscope equipped with cross polarised light. Use of transmitted VLM requires extraction of the putative starch from the artefact and mounting on slides to visualise grain features properly (Kooyman 2015).
- Starch granule features valuable to taxonomic identification include: size, three-dimensional shape of the granule, surface texture, position and features of hilum, fissures or grooves originating from hilum, lamellae visibility, facets, presentation of and rotation of extinction cross under cross polarised light, morphology of extinction cross (sharp or diffuse, wavy or straight arms), and presence of facets on the margins of granules (Croft 2012; García-Granero et al. 2016; Torrence and Barton 2006; Zarrillo and Kooyman 2006). The degree of specificity that can be attained in terms of taxon identification is variable with starch granules, depending on the quality of the reference collection consulted, and how much morphological and size overlap is present between species.
- When distinctive starch grain features are no longer visible, a destructive test involving digestion of the starch by a starch specific enzyme, such as α-amylase (Hardy et al. 2009) can confirm the presence of starch. Alternatively, a bath in a weak solution of hydrochloric acid can clarify if the putative starch granules are calcium carbonate formations, such as calcareous spherulites.
- It is important to note that the presence of only a few starch granules on an artefact might represent ancient incidental contact or modern contamination, particularly since airborne starch travels readily (Dozier 2016), and can be a contaminant in modern environments, including laboratories (Crowther et al. 2014).
- Potential starch granules are more easily spotted on smooth surfaces, such as fine-grained flint or obsidian.
Reference residue
- During reflected VLM observation of the reference potato residue, round and ovate objects that displayed partial extinction crosses in cross polarised light were found. These objects are presumably starch granules (Figure 31).
- No lamellae, hila, vacuoles, or other features were visible with SEM. However, the three-dimensional shape of the granules was better delineated by the SEM (Figure 32). SEM may be helpful in cases where the 3D shape of the granules are difficult to capture with reflected and transmitted VLM, for instance lenticular shapes that settle on slides in the flat orientation (polar view). The 3D shape of starch granules is routinely examined by applying a small amount of pressure on the coverslip of the slide with a probe, for example with an acupuncture needle or wood skewer. This procedure is only applicable to certain types of slide mounting media (e.g. water and glycerol) and thus ineffective with in situ starch on stone.


Wood
Biological description
- Different levels of identification can be attained with microscopic wood remains, from generic 'wood' down to individual species, depending on the number of features that are visible and the microscopic techniques used. In-depth description of wood anatomy is beyond the scope of this article, but in general conifer wood (softwood) exhibits less overall morphological variability among cells and shows simpler structure than dicotyledonous wood (hardwoods).
- Bordered pits are openings in the lignified secondary cell walls of tracheids and vessels, and are the major conduit through which water and solutes are transported in higher plants (Choat et al. 2008, 608).
Identification
- When viewed under transmitted cross polarised light, bordered pits display extinction crosses, which will rotate when the polarizing filter is rotated. This positive birefringence is due to the arrangement of the cellulose microfibrils in the bordered pits (Wayne 2009, 376).
- In conifers, the pits of tracheids are circular or slightly elliptical in plan view, large (usually 10-20 μm), and have wide borders that are raised above the level of the inner secondary wall, with a torus in the centre of the pit opening (Evert 2006b, 260). Across the bordered pit aperture, there is a membrane that has a round torus thickening in the centre, a feature characteristic of softwoods (Wilson and White 1986, 56). Unfortunately, the torus is not visible with reflected VLM when tracheids are viewed in situ on the artefact, necessitating the use of SEM.
- To distinguish between tracheids of conifers and vessel elements of deciduous trees/herbaceous plants, additional microscopy beyond reflected VLM may be required, either by SEM or extraction and mounting on slides and viewing with transmitted VLM. Transmitted VLM offers the advantage of allowing the examination of the internal structure of the vascular tissue cells, which may contain key features. For instance, the inner face of the lignified secondary wall of tracheids is sometimes lined with spiral thickenings, annular rings, and other patterns of taxonomic discrimination value.
- The diameter of pits in softwood and hardwood cells can be measured as a further aid to species level identification (Panshin and de Zeeuw 1980, 405–666).
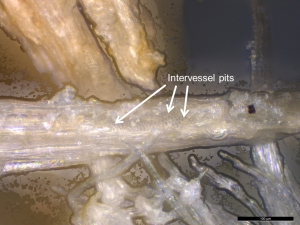
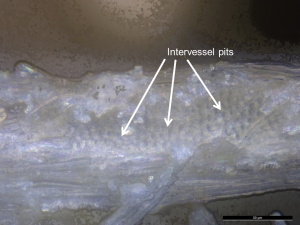
- The shape, number, and arrangement of pits on the side walls of tracheids and vessel elements can also be used for taxonomic discrimination. For instance, hardwood vessels display the following arrangements: alternate intervessel pitting (see Figures 35 and 36), opposite pitting, or scalariform pitting (Panshin and de Zeew 1980, 175), whereas bordered pits of softwood tracheids can be found arranged in uniseriate (linear single file), biseriate, or multiseriate rows along the tracheid length (Evans 2013, 167; Tyson 1995, 153).
- The structure of perforation plates in hardwood vessel elements (simple, scalariform, foraminate – also known as reticulate) can give important additional clues to wood residue taxon (Meylan and Butterfield 1972, 33).
Identification notes
- Apart from the lignified remains of tracheids, silica casts of these cells in the form of wood phytoliths may offer a route to identification. For instance, experimental research by Anderson (1980) suggests silica within wood cells can leave durable residues on lithic tool edges. Anderson also found the following wood cells were lined with silica: softwood tracheids, and hardwood vessel elements, fibres and parenchyma cells. Phytoliths from conifer tracheids maintaining bordered pits have been found preserved in two species of conifer needles by Bozarth (1993, 99), and almost all of the tracheid phytoliths from 42 species of conifer needles investigated by Hodson et al. (1997) and Sangster et al. (1997) had bordered pit impressions. Investigation of wood phytoliths and their taxonomic discrimination value is an area of inquiry that requires additional research, but appears to have potential (Piperno 2006, 42).
- Our finding that wood residues can only tentatively be identified when viewed in situ, is in agreement with Hardy and Garufi (1998, 180), who found that most experimental wood residues on stone tools do not have microscopically visible diagnostic features.
Reference residues
- Wood residues from a softwood (Figure 33) and hardwood (Figures 35 and 36) that were examined in situ on experimental flakes with reflected VLM lacked diagnostic anatomical features. However, many wood residues were distinctive in appearance to allow tentative, but not secure, identification. In particular the presence of bordered pits (structures that assist in the movement of water, nutrients and solutes through plants) are likely indicative of tool contact with woody plant matter.
- In the softwood, bordered pits were only found with SEM (Figure 34). In the hardwood, the presence of crystals, probably calcium oxalates, were detected and better visualisation of the pits was possible (Figure 37).



Red ochre
Description
- Ochre is a natural earth pigment that is comprised of clay, quartz, and iron oxides or oxyhydroxides (Dayet et al. 2013, 3492). In archaeology, ochre is used in a broader sense to mean any mineral material containing iron oxide (Mortimore et al. 2004, 1179) that can be used for colouring by leaving a red or yellow mark when rubbed against a surface.
- Red ochre takes its reddish colour from the mineral hematite (α-Fe2O3), which is dehydrated iron oxide. The hematite mineral itself is hexagonal (Rafferty 2012, 267), but red ochre occurs as nodules in nature.
Identification
- Microscopically, powdered red ochre appears granular.
- Ochre is described by Langejans and Lombard (2015, 210) as granular and appearing dull in cross-polarised light.
- Gibson et al. (2004, 3), describes ochre residues on stone tools as iron oxides that have grain sizes smaller than sand and can be dark red to mustard yellow.
- Although red ochre is distinctive in many circumstances, chemical methods are needed to confirm its presence. SEM-EDS provides a method for determining the presence of iron in suspected red ochre residue.
Reference residue
- The red ochre reference residue can be described as a distinctive red powdery material that is granular in texture and has a small grain size, in this case about 2-3 μm (Figure 38). However, there were no diagnostic characteristics detected using reflected VLM and there are a number of iron-based soil contaminants that are also red and granular.
- During SEM imaging, the ochre residue appeared as dark accumulations within depressed areas of the flint microtopography (Figure 39).

