Smirnou and Rehren (2011, 62) refer to the cobalt-blue frit found by Petrie and studied by Shortland and Tite (Tite et al. 1998; Shortland 2000; Shortland and Tite 2000; Tite and Shortland 2003; Shortland et al. 2006a) and Jackson and Nicholson (2007) as 'enigmatic', with its presence underlying 'the possible existence of local glass-making, but also the potentially complex nature of the materials and processes involved in LBA glass-making, as indicated by the cuneiform texts (Oppenheim et al. 1970)'. However, if we consider the cobalt-blue frit found at Amarna O45.1 as a vitreous concentrated colourant to be added to base glasses or their precursor materials, we potentially have the key to discovering the complex nature of these materials and processes.
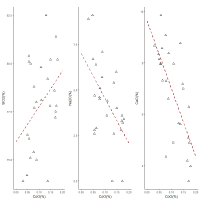
If the concentrated cobalt frit from Amarna and base glass mixture potentially provided the initial composition of the cobalt-blue glass that was used for some objects, any subsequent dilutions should be reflected in plots of the main compositional components of glass against the CoO concentration. The pXRF analyses on 26 blue glass samples from the 18th Dynasty plotted in Figure 6 present the opportunity to examine this. Although it is recognised that pXRF is a surface technique and that soluble species are more likely to be depleted at the surface (i.e. leaching of alkali oxides), the overall trends with CoO support the introduction of cobalt via a frit.


Despite the scatter in the data, Figure 18 shows that the silica concentration is lower at lower concentrations of CoO. Moreover, there is potentially a bimodal distribution, with the higher SiO2 concentrations perhaps reflecting the presence of unreacted quartz from the frit entering the glass, as found in the cobalt-blue frit at Amarna site 045.1 and the shoulder on the SiO2 distribution potentially reflecting the subsequent dilution with a base glass of lower SiO2 concentration. The CaO and Na2O concentrations appear to increase with decreasing CoO concentration. The CaO also exhibits a bimodal distribution, which suggests that the initial glass (shoulder) had a lower CaO concentration because it was a mixture of frit (with low CaO) with a base glass, followed by dilution with a base glass with higher CaO concentration. The Na2O level is quite low (Na2O~3%) which is probably a consequence of the pXRF detecting leaching at the surface (or the limits of pXRF to detect sodium even under vacuum conditions). Na2O also does not exhibit a bimodal distribution, although the peak is non-symmetrical and there is possible evidence of a shoulder. Nevertheless, the analysis is consistent with the hypothesis that the initial glass derived from mixing concentrated cobalt in the form of a frit (with low soda, low lime and high silica) to a base glass.
In essence, the plots in Figure 18 support the premise that a base glass was mixed with cobalt glass frit (with higher silica, lower soda, lower lime and concentrated cobalt) to produce the initial blue glass used for objects, which was later diluted with a base glass in the 18th Dynasty and again in the Ramesside period. Assuming that the 18th Dynasty dilution is the result of mixing blue glass with 5%CaO (shoulder) and 78%SiO2 (upper peak) in a ratio of 1:1 with a base glass, suggests that the base glass would have had a CaO concentration of 11% and a SiO2 concentration of 72%. These are reasonable values for a stable base glass. In summary, based on the pXRF data from Abe et al. (2012), 18th Dynasty cobalt-blue glass had an initial average composition of around 78%SiO2, 5%CaO, 0.16%CoO, 0.11%NiO, 0.38%MnO and about 0.15%ZnO and 3.9%Al2O3 (i.e. the higher peaks in Figure 13 and Figure 14; the higher peak for SiO2 and the shoulder for CaO in Figure 18) prior to any dilution.
The mixing of a cobalt-concentrated frit with a base glass opens up the possibility that although the base glass may have been made locally, the concentrated frit colourant may have derived from elsewhere. On first inspection this would appear to contradict the work of Shortland et al. (2007) and their use of Ti, Cr, Zr and La to discriminate between Egyptian and Mesopotamian sources of glass. However, as shown in Figure 15, higher levels of Ti are associated with clays derived from Nile river silt. Similarly, elevated levels of La and Zr found in Egyptian glasses have been attributed to this Nile source, while elevated Cr levels found in Mesopotamian glasses have been attributed to alluvial soils derived from the weathering of ultramafic rocks in Turkey at the head-waters of the Tigris and Euphrates rivers (Shortland et al. 2007; Walton et al. 2009). Since these elements potentially enter the glass by the partial melting of the clay crucible (i.e. contaminating the glass in a similar way to Al2O3), they will yield a signature of the location where the colourant was added to the base glass, and not necessarily the location from where the colourant derived. The same rationale can be applied to the Sr and Nd isotope analyses conducted on glasses from Egypt and the Near East by Degryse et al. (2010), which can identify the base glass precursors and not necessarily the additives. In other words, these studies do not rule out a Bronze Age trade network of a vitreous cobalt frit colourant, in a similar fashion to how smalt was traded to China from Europe in the Qing Dynasty period for porcelain (Giannini et al. 2017).
Internet Archaeology is an open access journal based in the Department of Archaeology, University of York. Except where otherwise noted, content from this work may be used under the terms of the Creative Commons Attribution 3.0 (CC BY) Unported licence, which permits unrestricted use, distribution, and reproduction in any medium, provided that attribution to the author(s), the title of the work, the Internet Archaeology journal and the relevant URL/DOI are given.
Terms and Conditions | Legal Statements | Privacy Policy | Cookies Policy | Citing Internet Archaeology
Internet Archaeology content is preserved for the long term with the Archaeology Data Service (ROR). Help sustain and support open access publication by donating to our Open Access Archaeology Fund.